Description
PanBRCA™ Inherited Breast & Ovarian Cancer Screening Test
PanBRCA™ (OBS) : $8,160
PanBRCA™ (OBF) : $15,600
Breast cancer is the third most deadly cancer among women in Hong Kong, second only to lung cancer and colorectal cancer. In recent years, many studies have found that mutations in BRCA1&2 genes are related to various cancers. The occurrence of pathogenic mutations in BRCA genes will lead to the failure of the DNA repair mechanism and increase the risk of cancers. If there is an inherited mutation in genes, the chance of rapid cell division and change increases, and the possibility of developing cancer is also greater.
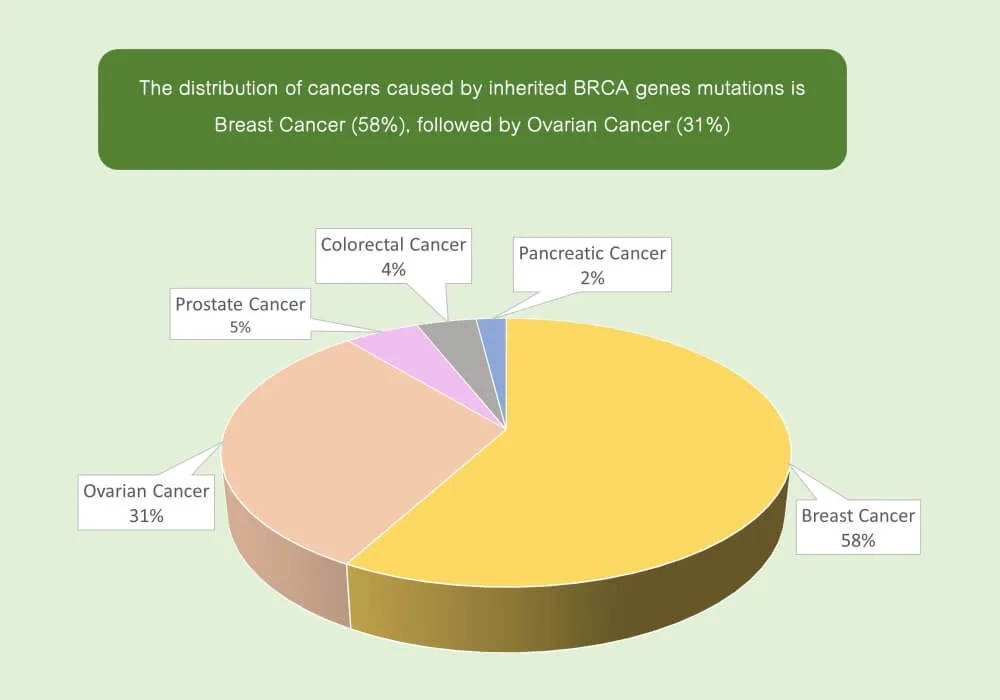
- In 2019, a total of 852 women died from this cancer in Hong Kong, accounting for 13.7% of all cancer deaths in females
- It is estimated that more than 5 million people in China carry BRCA disease-causing gene mutations
- In Asians, the risk of a cancer-causing mutation in the BRCA gene represent as below: 87% of Breast cancer
- 44% of ovarian cancer and also an increased risk for other types of cancer (such as pancreatic cancer, colorectal cancer, prostate cancer, etc.)
Specifically Designed for Asians – Common Asian – Specific Gene Mutations
- Using next-generation sequencing (NGS) and the most advanced technology to detect any genetic mutations, including gene rearrangements and large deletions of BRCA1&2 genes, related to hereditary breast cancer and ovarian cancer
- Designed for screening Asian genetic mutations, with coverage up to 95%
High Accuracy Trustworthy Bioinformatics
- PanBRCA™ uses advanced data analysis technology to achieve a higher detection and analytical sensitivity than most other BRCA test in the market
- A confirmatory test will be performed in dual to ensure the highest accuracy
- The test is highly rated with exceptional performance in the Proficiency testing program provided by the College of American Pathologist (CAP)
PanBRCA™ Screening Test Features
| Other Tests (HRM) | PanBRCA™ (OBS) | PanBRCA™ (OBF) | |
| Price | – | $8,160 | $15,600 |
| BRCA genes Mutation Coverage % | 73-90% | Up to 90-95% | Up to 99.9% |
| Turnaround time (TAT) | Irregular | Within 14-20working days | |
| Superiority | – | Golden Standard & Comprehensive Coverage | |
Test Specifications
| Test Code | Methodology | Specimen Requirements |
| OBS | NGS (Next Generation Sequencing) | 6ml of EDTA blood / 10 Buccal Swabs |
| OBF | NGS + Multiplex ligation-dependent probe amplification (MLPA) |
PanBRCA™ Screening Test
- Collect samples by drawing 6ml of EDTA blood or 10 buccal swabs
- Within 14-20 working days
- 100% Hong Kong laboratory, analysis and professional test report
Suitable for
- Early onset breast cancer (<45years old)
- Bilateral or multiple primary breast cancers
- Male breast cancer at any age
- Breast and ovarian cancer in the same women
- 3 or more cases of breast cancer in the family
- 3 or more cases of breast, ovarian and/or pancreatic cancer in the family
- 3 or more cases of breast, uterine and/or thyroid cancer in the family
- Multiple close family members with breast and other cancers
Instructions for use:
- Must be used on or before the expiration date
- Please show your confirmation information, order number, registered full name and phone number for verification
Booking Process and Terms & Conditions:
- Customers will receive a confirmation email for successful payment after purchasing a gene test from the Trinity Medical Centre online store. Trinity Medical Centre will contact customers during subsequent business hours to schedule the time and location for the gene test appointment. Customers may also contact us for an appointment after order confirmation (Tel: 2192 7022 or Email: tmc@trinitymedical.com.hk).
- This package is exclusively available on Trinity Medical Centre’s online store and must be conducted at designated Trinity Medical Centres. It cannot be used with other promotions. The offer is non-transferable, non-refundable, and cannot be exchanged for full or partial cash value.
- Customers must schedule and undergo the relevant examination within 90 days of purchase; the offer becomes void after this date.
- If this voucher is lost or stolen, we will not accept any liability and will not provide any reissue in such case.
- All customers must present identification documents and the successful payment confirmation email during registration at the centre to be eligible for the service.
- If a parent or guardian purchases the service for a minor, the service user must accompany the parent or guardian to the centre for the examination.
- For any inquiries, please call 2192 7022 or email tmc@trinitymedical.com.hk.
- Trinity Medical Centre reserves the right to amend the terms and conditions without prior notice.
- In case of any disputes, Trinity Medical Centre reserves the right of final decision.















